Background: Chimeric antigen receptor T-cell (CAR-T) therapy is an effective strategy for patients (pts) with B-cell non-Hodgkin lymphomas (B-NHLs). However, long-term remissions are only observed in ~40% of pts with large cell lymphomas (LCL) treated with FDA-approved CD19-targeted products and there is currently no approved CAR-T for other histologies other than mantle cell lymphoma (MCL). CD20 is a proven therapeutic target for B-NHL, supported by previously approved naked and radiolabeled anti-CD20 antibodies and promising results from bispecific antibodies. CD20-targeted CAR-T is another potential adoptive immunotherapy option that could be utilized in combination or in sequence with CD19 CAR-T. We present interim results of our ongoing phase I/II clinical trial investigating safety and efficacy CD20 CAR-T for high-risk B-NHLs (NCT03277729).
Methods: MB-106 is a fully human third-generation CD20-targeted CAR-T with both 4-1BB and CD28 costimulatory domains. We use a continuous reassessment method dose escalation design to find the maximally tolerated dose. Lymphodepletion (LD) consists of cyclophosphamide (Cy) ± fludarabine (Flu). CAR-T cells are administered at one of 4 dose levels (DL): DL1: 3.3 x 105, DL2: 1 x106, DL3: 3.3x106, DL4: 1x107 CAR T cells/kg. CAR-T is infused in the outpatient setting except for the first pts of each dose cohort (overnight observation). Pts with CD20+ B-NHL are eligible, including but not limited to LCL, follicular lymphoma (FL)/indolent histologies and MCL. Prior treatment with CD19 CAR-T is permitted. Treatment response is assessed on day 28 using 2014 Lugano criteria. Cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) were graded per 2014 Lee criteria and the ASTCT consensus grading, respectively.
Results: Between 2018-2020, 12 pts underwent leukapheresis (LA) and 11 (92%) were treated on the protocol. The first 7 pts (3 FL, 3 MCL, 1 hairy cell variant) were treated using the original cell manufacturing method with separate culturing of CD4+ and CD8+ cells ("original process"). First 4 of those 7 pts (2 treated at DL1 and 2 at DL2) only received Cy for LD and the other 3 (all at DL1) were treated after LD with Cy-Flu. There was no evidence of ICANS in those 7 pts and only 1 pt developed CRS (grade 3 - unexplained elevated alkaline phosphatase (ALP) in the setting of fever). Responses at 4 weeks for pts treated with the "original process" included stable disease (SD) in 4 and progressive disease (PD) in 3 pts. Given the challenges in meeting the protocol requirements for CD4+ and CD8+ specific doses, poor CAR-T expansion, and lack of clinical responses, enrollment was placed on a hold and cell manufacturing process underwent a major revision which included changing to a combined culture of CD4+ and CD8+ cells, among other modifications ("modified process").
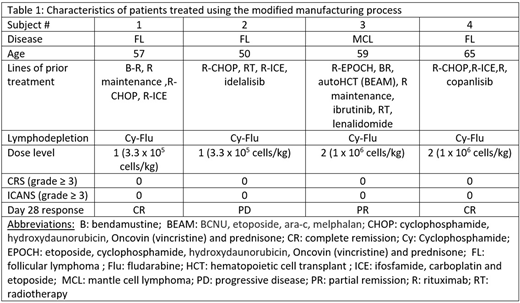
Since December 2019 enrollment was reinitiated at DL1 under the "modified process." Four pts were treated after LD with Cy-Flu at DL1 (2 FL) and dose level 2 (1 FL and 1 MCL) (details in Table 1). Target cell doses were achieved in all 4 patients and treatment was tolerated with no occurrence of CRS (any grade) or ICANS (any grade). Responses were observed at both dose levels and included complete remissions (CRs) in 2 FL pts (1 treated at DL1 and 1 at DL2), partial remission (PR) in a MCL pt treated at DL2 and PD in a FL pt who was treated at DL1. Robust CAR-T expansion was seen in 3 of the 4 patients, with peak blood levels of CAR+ T cells of 161, 5.5, and 401 CAR+ cells/µl, corresponding to 80%, 23%, and 72% of all CD8+ cells.
Combining all 11 pts treated under the "original" and "modified" processes, no dose-limiting toxicity (DLT) was observed. Grade ≥ 3 toxicities included: anemia (n=4, 36%), lymphopenia (n=3,27%), febrile neutropenia (n=2, 18%), hypertension (n=1, 9%) , hypotension (n=1, 9%), thromboembolic event (n=1, 9%), neutropenia (n=1, 9%), elevated ALP (n=1, 9%), pneumonia (n=1, 9%), bacteremia (n=1, 9%), hyperglycemia (n=1, 9%), pleural effusion (n=1, 9%) and generalized pain (n=1, 9%).
Conclusion: Early results of our ongoing study of CD20 CAR-T for high-risk NHLs suggests an extremely favorable safety profile (no CRS or ICANS of any grades) with the modified manufacturing process. There is also evidence of clinical activity even at low dose levels with observed CRs at DLs 1 and 2. Enrollment at higher DLs is currently ongoing and the data will be updated at the time of presentation.
Shadman:Abbvie, Genentech, Astra Zeneca, Sound Biologics , Pharmacyclics, Verastem, ADC therapeutics, Beigene, Cellectar, BMS, Morphosys and Atara Biotherapeutics: Consultancy; Mustang Bio, Celgene, Pharmacyclics, Gilead, Genentech, Abbvie, TG therapeutics, Beigene, Astra Zeneca, Sunesis, Beigene: Research Funding. Yeung:OBI;Pfizer: Research Funding; Adaptive Biotechnology;Eli Lilly;Merck: Consultancy. Ramachandran:Mustang Bio: Current Employment. Graf:TG Therapeutics: Research Funding; BeiGene: Research Funding; MorphoSys: Consultancy; Acerta Pharma: Research Funding. Gopal:IgM bio, BMS, merck: Research Funding; Seattle Genetics; Janssen; IMab Bio; TG Therapeutics; Astra Zeneca; Merck; Gilead; ADC Therapeutics; Nurix; TG therapeutics, Cellectar; Actinium: Consultancy; imab bio, takeda,astrazeneca,gilead: Research Funding; Seattle Genetics; Janssen; Takeda; IgM Bio; IMab Bio; BMS; Astra Zeneca; Merck; Gilead: Research Funding. Gauthier:JMP, Eusapharma, Multerra Bio: Honoraria. Cassaday:Pfizer: Honoraria, Research Funding; Seattle Genetics: Current Employment, Current equity holder in publicly-traded company; Vanda Pharmaceuticals: Research Funding; Kite/Gilead: Consultancy, Research Funding; Merck: Research Funding; Amgen: Consultancy, Research Funding. Kiem:Magenta Therapeutics, CSL,Homology Medicines, Vor Biopharma , Enochian, Umoja, Rocket Pharma: Consultancy. Turtle:Novartis: Consultancy; Myeloid Therapeutics: Current equity holder in private company, Honoraria, Membership on an entity's Board of Directors or advisory committees; Allogene: Consultancy; PACT Pharma: Consultancy; Arsenal Bio: Current equity holder in private company, Honoraria, Membership on an entity's Board of Directors or advisory committees; Caribou Biosciences: Current equity holder in private company, Honoraria, Membership on an entity's Board of Directors or advisory committees; Eureka Therapeutics: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Precision Biosciences: Current equity holder in publicly-traded company, Honoraria, Membership on an entity's Board of Directors or advisory committees; Physician Education Resource: Consultancy; AstraZeneca: Consultancy, Research Funding; Nektar Therapeutics: Consultancy, Research Funding; Juno/BMS: Patents & Royalties, Research Funding; Century Therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees; T-CURX: Membership on an entity's Board of Directors or advisory committees; Humanigen: Consultancy; Kite/Gilead: Consultancy. Maloney:Pharmacyclics: Consultancy, Honoraria; Juno Therapeutics: Consultancy, Honoraria, Patents & Royalties: Patents are pending, but not issued, licensed, no royalties, no licensees., Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Kite, a Gilead Company: Consultancy, Honoraria, Research Funding; Gilead Sciences: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; MorphoSys: Consultancy, Honoraria; Bioline Rx: Consultancy, Honoraria; A2 Biotherapeutics: Consultancy, Current equity holder in publicly-traded company, Honoraria; Genentech: Consultancy, Honoraria. Till:Mustang: Patents & Royalties, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal